
SOL.i.PHARMA certifications ensure quality, safety, and full regulatory compliance in the pharmaceutical and medical sector. Each system is developed according to international standards and validated to operate in controlled environments.
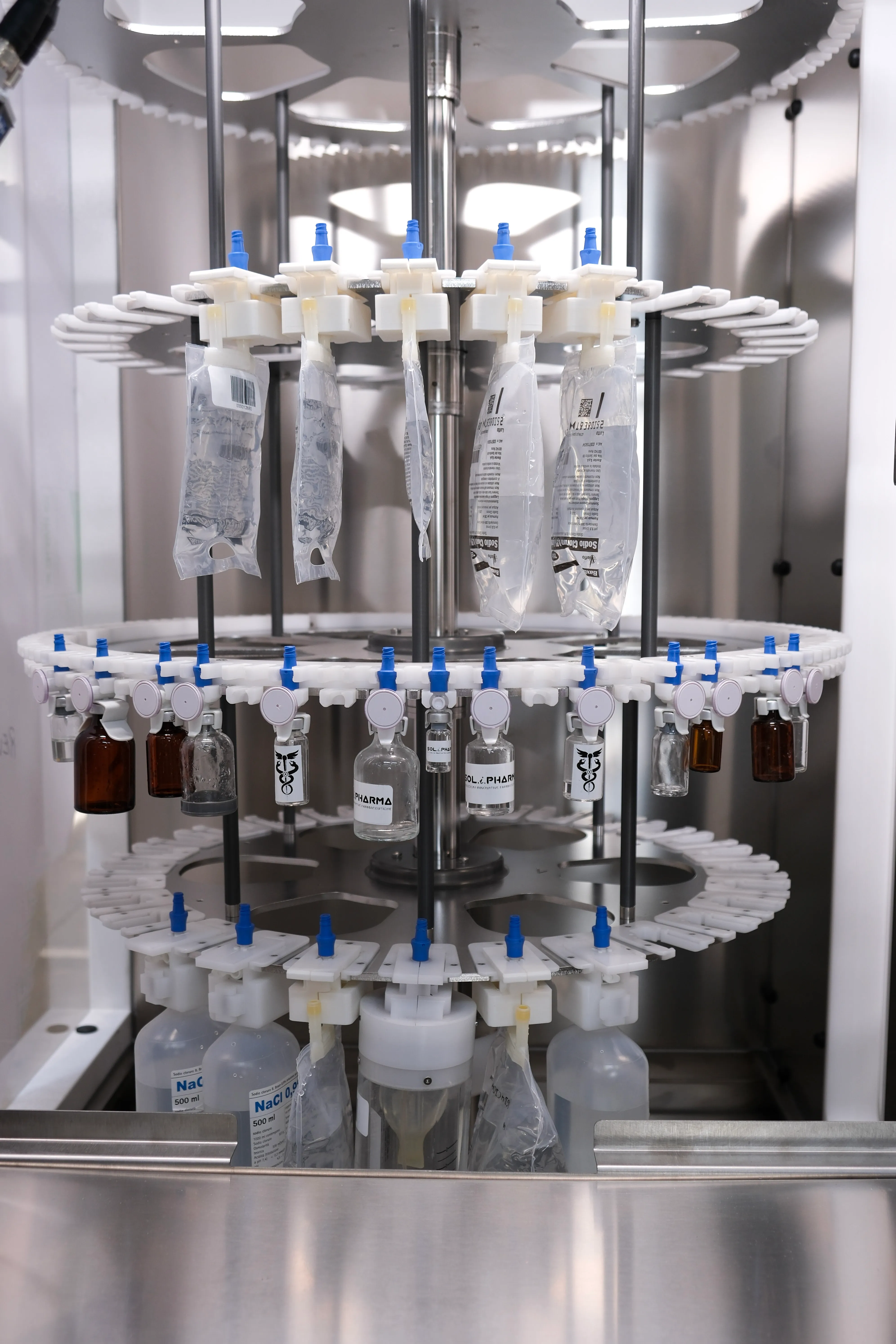
Our systems are designed to ensure operational safety, regulatory compliance, and consistent performance in highly regulated pharmaceutical and medical environments.

Each system is developed and verified through a structured process that ensures regulatory compliance, consistent quality, and operational safety.
SOL.i.PHARMA is certified according to ISO 9001 standards for quality management. Additionally, its systems comply with applicable European CE directives and are tested for use in controlled environments.
CE compliance indicates that the systems meet European regulations regarding safety, performance, and reliability. This includes directives such as the Machinery Directive and electromagnetic compatibility.
Yes, the systems are tested for controlled environments and are compatible up to specific ISO classes, ensuring safety and control of particulate emissions.
Through ISO 9001 certified systems, we adopt structured processes for quality control, continuous improvement, and operational traceability.
Yes, certifications are maintained and periodically renewed to ensure compliance with updated regulations and industry standards.
Yes, certificates and declarations of conformity are available for download directly in the dedicated section of the page.
Discover how to integrate automation into your hospital pharmacy and improve safety, precision, and control in the preparation of antiblastic drugs.